Search for drugs:
Typing the drug name to query
ATORVASTATIN CALCIUM
DIR Classification
Classification:Most-DIR concern
Severity Score:4
Description in Drug Labeling: View Full Labeling: SPL in DailyMed | PDF
- WARNINGS AND PRECAUTIONS
- Skeletal Muscle
- Rare cases of rhabdomyolysis with acute renal failure secondary to myoglobinuria have been reported with atorvastatin calciumand with other drugs in this class. A history of renal impairment may be a risk factor for the development of rhabdomyolysis. Such patients merit closer monitoring for skeletal muscle effects.
- Atorvastatin, like other statins, occasionally causes myopathy, defined as muscle aches or muscle weakness in conjunction with increases in creatine phosphokinase (CPK) values > 10 times ULN. The concomitant use of higher doses of atorvastatin with certain drugs such as cyclosporine and strong CYP3A4 inhibitors (e.g., clarithromycin, itraconazole, and HIV protease inhibitors) increases the risk of myopathy/rhabdomyolysis.
- There have been rare reports of immune-mediated necrotizing myopathy (IMNM), an autoimmune myopathy, associated with statin use. IMNM is characterized by: proximal muscle weakness and elevated serum creatine kinase, which persist despite discontinuation of statin treatment; muscle biopsy showing necrotizing myopathy without significant inflammation; improvement with immunosuppressive agents.
- Myopathy should be considered in any patient with diffuse myalgias, muscle tenderness or weakness, and/or marked elevation of CPK. Patients should be advised to report promptly unexplained muscle pain, tenderness, or weakness, particularly if accompanied by malaise or fever or if muscle signs and symptoms persist after discontinuing atorvastatin calcium. Atorvastatin calcium therapy should be discontinued if markedly elevated CPK levels occur or myopathy is diagnosed or suspected.
- The risk of myopathy during treatment with drugs in this class is increased with concurrent administration of cyclosporine, fibric acid derivatives, erythromycin, clarithromycin, the hepatitis C protease inhibitor telaprevir, combinations of HIV protease inhibitors, including saquinavir plus ritonavir, lopinavir plus ritonavir, tipranavir plus ritonavir, darunavir plus ritonavir, fosamprenavir, and fosamprenavir plus ritonavir, niacin, or azole antifungals. Physicians considering combined therapy with atorvastatin calcium and fibric acid derivatives, erythromycin, clarithromycin, a combination of saquinavir plus ritonavir, lopinavir plus ritonavir, darunavir plus ritonavir, fosamprenavir, or fosamprenavir plus ritonavir, azole antifungals, or lipid-modifying doses of niacin should carefully weigh the potential benefits and risks and should carefully monitor patients for any signs or symptoms of muscle pain, tenderness, or weakness, particularly during the initial months of therapy and during any periods of upward dosage titration of either drug. Lower starting and maintenance doses of atorvastatin should be considered when taken concomitantly with the aforementioned drugs (see Drug Interactions(7)). Periodic creatine phosphokinase (CPK) determinations may be considered in such situations, but there is no assurance that such monitoring will prevent the occurrence of severe myopathy.
- Prescribing recommendations for interacting agents are summarized in Table 1 [see also Dosage and Administration (2.6), Drug Interactions (7), Clinical Pharmacology (12.3)].
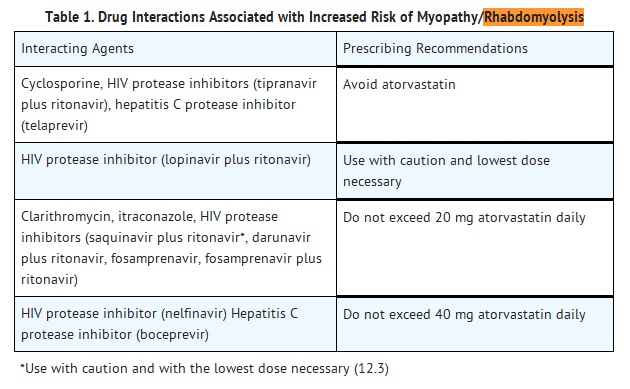
- Cases of myopathy, including rhabdomyolysis, have been reported with atorvastatin co-administered with colchicine, and caution should be exercised when prescribing atorvastatin with colchicine [see Drug Interactions (7.11)].
- Atorvastatin calcium therapy should be temporarily withheld or discontinued in any patient with an acute, serious condition suggestive of a myopathy or having a risk factor predisposing to the development of renal failure secondary to rhabdomyolysis (e.g., severe acute infection, hypotension, major surgery, trauma, severe metabolic, endocrine and electrolyte disorders, and uncontrolled seizures).
- ADVERSE REACTIONS
- Clinical Trial Adverse Experiences
- Because clinical trials are conducted under widely varying conditions, the adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
- In the atorvastatin calcium placebo-controlled clinical trial database of 16,066 patients (8755 atorvastatin calcium vs. 7311 placebo; age range 10 to 93 years, 39% women, 91% Caucasians, 3% Blacks, 2% Asians, 4% other) with a median treatment duration of 53 weeks, 9.7% of patients on atorvastatin calcium and 9.5% of the patients on placebo discontinued due to adverse reactions regardless of causality. The five most common adverse reactions in patients treated with atorvastatin calcium that led to treatment discontinuation and occurred at a rate greater than placebo were: myalgia (0.7%), diarrhea (0.5%), nausea (0.4%), alanine aminotransferase increase (0.4%), and hepatic enzyme increase (0.4%).
- The most commonly reported adverse reactions (incidence ≥ 2% and greater than placebo) regardless of causality, in patients treated with atorvastatin calcium in placebo controlled trials (n = 8755) were: nasopharyngitis (8.3%), arthralgia (6.9%), diarrhea (6.8%), pain in extremity (6%), and urinary tract infection (5.7%).
- Other adverse reactions reported in placebo-controlled studies include:
- Body as a whole: malaise, pyrexia; Digestive system: abdominal discomfort, eructation, flatulence, hepatitis, cholestasis; Musculoskeletal system: musculoskeletal pain, muscle fatigue, neck pain, joint swelling; Metabolic and nutritional system: transaminases increase, liver function test abnormal, blood alkaline phosphatase increase, creatine phosphokinase increase, hyperglycemia; Nervous system: nightmare; Respiratory system: epistaxis; Skin and appendages: urticaria; Special senses: vision blurred, tinnitus; Urogenital system: white blood cells urine positive.
- Anglo-Scandinavian Cardiac Outcomes Trial (ASCOT)
- In ASCOT [see Clinical Studies (14.1)] involving 10,305 participants (age range 40 to 80 years, 19% women; 94.6% Caucasians, 2.6% Africans, 1.5% South Asians, 1.3% mixed/other) treated with atorvastatin calcium 10 mg daily (n = 5,168) or placebo (n = 5,137), the safety and tolerability profile of the group treated with atorvastatin calcium was comparable to that of the group treated with placebo during a median of 3.3 years of follow-up.
- Collaborative Atorvastatin Diabetes Study (CARDS)
- In CARDS [see Clinical Studies (14.1)] involving 2,838 subjects (age range 39 to 77 years, 32% women; 94.3% Caucasians, 2.4% South Asians, 2.3% Afro-Caribbean, 1% other) with type 2 diabetes treated with atorvastatin calcium 10 mg daily (n = 1,428) or placebo (n = 1,410), there was no difference in the overall frequency of adverse reactions or serious adverse reactions between the treatment groups during a median follow-up of 3.9 years. No cases of rhabdomyolysis were reported.
- Postmarketing Experience
- The following adverse reactions have been identified during postapproval use of atorvastatin calcium. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Adverse reactions associated with atorvastatin calcium therapy reported since market introduction, that are not listed above, regardless of causality assessment, include the following: anaphylaxis, angioneurotic edema, bullous rashes (including erythema multiforme, Stevens-Johnson syndrome, and toxic epidermal necrolysis), rhabdomyolysis, fatigue, tendon rupture, fatal and non-fatal hepatic failure, dizziness, depression, peripheral neuropathy, and pancreatitis.
- There have been rare reports of immune-mediated necrotizing myopathy associated with statin use [see Warnings and Precautions (5.1)].
- There have been rare postmarketing reports of cognitive impairment (e.g., memory loss, forgetfulness, amnesia, memory impairment, confusion) associated with statin use. These cognitive issues have been reported for all statins. The reports are generally nonserious, and reversible upon statin discontinuation, with variable times to symptom onset (1 day to years) and symptom resolution (median of 3 weeks).
- DRUG INTERACTIONS
- The risk of myopathy during treatment with statins is increased with concurrent administration of fibric acid derivatives, lipid-modifying doses of niacin, cyclosporine, or strong CYP 3A4 inhibitors (e.g., clarithromycin, HIV protease inhibitors, and itraconazole) [see Warnings and Precautions, Skeletal Muscle (5.1) and Clinical Pharmacology (12.3)].
- Gemfibrozil
- Due to an increased risk of myopathy/rhabdomyolysis when HMG-CoA reductase inhibitors are co-administered with gemfibrozil, concomitant administration of atorvastatin calcium with gemfibrozil should be avoided [see Warnings and Precautions (5.1)].
- Colchicine
- Cases of myopathy, including rhabdomyolysis, have been reported with atorvastatin co-administered with colchicine, and caution should be exercised when prescribing atorvastatin with colchicine.
Postmarketing Surveillance
Contingency Table:
Current Drug
Other Drugs
Rhabdomyolysis
2973
39939
Other ADRs
78982
14038297
Odds Ratio = 13.231
Drug Property Information
ATC Code(s):
- C10AA05 - atorvastatin calcium
- C10AA - HMG CoA reductase inhibitors
- C10A - "LIPID MODIFYING AGENTS, PLAIN"
- C10 - LIPID MODIFYING AGENTS
- C - CARDIOVASCULAR SYSTEM
- C10BA05 - atorvastatin calcium
- C10BA - HMG CoA reductase inhibitors in combination with other lipid modifying agents
- C10B - "LIPID MODIFYING AGENTS, COMBINATIONS"
- C10 - LIPID MODIFYING AGENTS
- C - CARDIOVASCULAR SYSTEM
- C10BX06 - atorvastatin calcium
- C10BX - "HMG CoA reductase inhibitors, other combinations"
- C10B - "LIPID MODIFYING AGENTS, COMBINATIONS"
- C10 - LIPID MODIFYING AGENTS
- C - CARDIOVASCULAR SYSTEM
- C10BX11 - atorvastatin calcium
- C10BX - "HMG CoA reductase inhibitors, other combinations"
- C10B - "LIPID MODIFYING AGENTS, COMBINATIONS"
- C10 - LIPID MODIFYING AGENTS
- C - CARDIOVASCULAR SYSTEM
- C10BX03 - atorvastatin calcium
- C10BX - "HMG CoA reductase inhibitors, other combinations"
- C10B - "LIPID MODIFYING AGENTS, COMBINATIONS"
- C10 - LIPID MODIFYING AGENTS
- C - CARDIOVASCULAR SYSTEM
- C10BX08 - atorvastatin calcium
- C10BX - "HMG CoA reductase inhibitors, other combinations"
- C10B - "LIPID MODIFYING AGENTS, COMBINATIONS"
- C10 - LIPID MODIFYING AGENTS
- C - CARDIOVASCULAR SYSTEM
Active Ingredient:atorvastatin calcium
Active Ingredient UNII:48A5M73Z4Q
Drugbank ID:DB01076
PubChem Compound:60823
CAS Number:134523-00-5
Dosage Form(s):tablet, film coated
Route(s) Of Administrator:oral
Daily Dose:
- 20.0 mg/day C10AA05
Chemical Structure: 

SMILE Code:
CC(C)C1=C(C(=C(N1CC[C@H](C[C@H](CC(=O)O)O)O)C2=CC=C(C=C2)F)C3=CC=CC=C3)C(=O)NC4=CC=CC=C4
CC(C)C1=C(C(=C(N1CC[C@H](C[C@H](CC(=O)O)O)O)C2=CC=C(C=C2)F)C3=CC=CC=C3)C(=O)NC4=CC=CC=C4
Reference
COHORT STUDY:
1: Incidence of hospitalized rhabdomyolysis in patients treated with lipid-lowering drugs.
[Graham DJ, Staffa JA, Shatin D, Andrade SE, Schech SD, La Grenade L, Gurwitz JH, Chan KA, Goodman MJ, Platt R, JAMA. 2004 Dec 1;292(21):2585-90.]ABSTRACT
CONTEXT: Lipid-lowering agents are widely prescribed in the United States. Reliable estimates of rhabdomyolysis risk with various lipid-lowering agents are not available.
OBJECTIVE: To estimate the incidence of rhabdomyolysis in patients treated with different statins and fibrates, alone and in combination, in the ambulatory setting.
DESIGN, SETTING, AND PATIENTS: Drug-specific inception cohorts of statin and fibrate users were established using claims data from 11 managed care health plans across the United States. Patients with at least 180 days of prior health plan enrollment were entered into the cohorts between January 1, 1998, and June 30, 2001. Person-time was classified as monotherapy or combined statin-fibrate therapy.
MAIN OUTCOME MEASURE: Incidence rates of rhabdomyolysis per 10,000 person-years of treatment, number needed to treat, and relative risk of rhabdomyolysis.
RESULTS: In 252,460 patients treated with lipid-lowering agents, 24 cases of hospitalized rhabdomyolysis occurred during treatment. Average incidence per 10,000 person-years for monotherapy with atorvastatin, pravastatin, or simvastatin was 0.44 (95% confidence interval [CI], 0.20-0.84); for cerivastatin, 5.34 (95% CI, 1.46-13.68); and for fibrate, 2.82 (95% CI, 0.58-8.24). By comparison, the incidence during unexposed person-time was 0 (95% CI, 0-0.48; P = .056). The incidence increased to 5.98 (95% CI, 0.72-216.0) for combined therapy of atorvastatin, pravastatin, or simvastatin with a fibrate, and to 1035 (95% CI, 389-2117) for combined cerivastatin-fibrate use. Per year of therapy, the number needed to treat to observe 1 case of rhabdomyolysis was 22,727 for statin monotherapy, 484 for older patients with diabetes mellitus who were treated with both a statin and fibrate, and ranged from 9.7 to 12.7 for patients who were treated with cerivastatin plus fibrate.
CONCLUSIONS: Rhabdomyolysis risk was similar and low for monotherapy with atorvastatin, pravastatin, and simvastatin; combined statin-fibrate use increased risk, especially in older patients with diabetes mellitus. Cerivastatin combined with fibrate conferred a risk of approximately 1 in 10 treated patients per year.
PMID: 15572716
OBJECTIVE: To estimate the incidence of rhabdomyolysis in patients treated with different statins and fibrates, alone and in combination, in the ambulatory setting.
DESIGN, SETTING, AND PATIENTS: Drug-specific inception cohorts of statin and fibrate users were established using claims data from 11 managed care health plans across the United States. Patients with at least 180 days of prior health plan enrollment were entered into the cohorts between January 1, 1998, and June 30, 2001. Person-time was classified as monotherapy or combined statin-fibrate therapy.
MAIN OUTCOME MEASURE: Incidence rates of rhabdomyolysis per 10,000 person-years of treatment, number needed to treat, and relative risk of rhabdomyolysis.
RESULTS: In 252,460 patients treated with lipid-lowering agents, 24 cases of hospitalized rhabdomyolysis occurred during treatment. Average incidence per 10,000 person-years for monotherapy with atorvastatin, pravastatin, or simvastatin was 0.44 (95% confidence interval [CI], 0.20-0.84); for cerivastatin, 5.34 (95% CI, 1.46-13.68); and for fibrate, 2.82 (95% CI, 0.58-8.24). By comparison, the incidence during unexposed person-time was 0 (95% CI, 0-0.48; P = .056). The incidence increased to 5.98 (95% CI, 0.72-216.0) for combined therapy of atorvastatin, pravastatin, or simvastatin with a fibrate, and to 1035 (95% CI, 389-2117) for combined cerivastatin-fibrate use. Per year of therapy, the number needed to treat to observe 1 case of rhabdomyolysis was 22,727 for statin monotherapy, 484 for older patients with diabetes mellitus who were treated with both a statin and fibrate, and ranged from 9.7 to 12.7 for patients who were treated with cerivastatin plus fibrate.
CONCLUSIONS: Rhabdomyolysis risk was similar and low for monotherapy with atorvastatin, pravastatin, and simvastatin; combined statin-fibrate use increased risk, especially in older patients with diabetes mellitus. Cerivastatin combined with fibrate conferred a risk of approximately 1 in 10 treated patients per year.
OTHER REFERENCE(S):
1: Rhabdomyolysis-induced compartment syndrome secondary to atorvastatin and strenuous exercise.
[Dunphy Louise,Morhij Rossel,Tucker Sarah]BMJ Case Rep.2017 Mar 16;2017. pii: bcr2016218942. doi: 10.1136/bcr-2016-218942. PMID: 28302660
2: Results of a Doravirine-Atorvastatin Drug-Drug Interaction Study.
[Khalilieh Sauzanne,Yee Ka Lai,Sanchez Rosa I,Triantafyllou Ilias,Fan Li,Maklad Noha,Jordan Heather,Martell Maureen,Iwamoto Marian]Antimicrob Agents Chemother.2017 Jan 24;61(2). pii: e01364-16. doi: 10.1128/AAC.01364-16. Print 2017 Feb. PMID: 27872071
3: Appropriate risk criteria for OATP inhibition at the drug discovery stage based on the clinical relevancy between OATP inhibitors and drug-induced adverse effect.
[Nakakariya Masanori,Goto Akihiko,Amano Nobuyuki]Drug Metab Pharmacokinet.2016 Oct;31(5):333-339. doi: 10.1016/j.dmpk.2016.05.003. Epub 2016 May 28. PMID: 27567380
4: Safety of atorvastatin in Asian patients within clinical trials.
[Chan Juliana C N,Kong Alice P S,Bao Weihang,Fayyad Rana,Laskey Rachel]Cardiovasc Ther.2016 Dec;34(6):431-440. doi: 10.1111/1755-5922.12214. PMID: 27520479
5: Rhabdomyolysis caused by the moderate CYP3A4 inhibitor fluconazole in a patient on stable atorvastatin therapy: a case report and literature review.
[Hsiao S-H,Chang H-J,Hsieh T-H,Kao S-M,Yeh P-Y,Wu T-J]J Clin Pharm Ther.2016 Oct;41(5):575-8. doi: 10.1111/jcpt.12425. Epub 2016 Jul 19. PMID: 27430348
6: Comparative Effectiveness of Generic Atorvastatin and Lipitor® in Patients Hospitalized with an Acute Coronary Syndrome.
[Jackevicius Cynthia A,Tu Jack V,Krumholz Harlan M,Austin Peter C,Ross Joseph S,Stukel Therese A,Koh Maria,Chong Alice,Ko Dennis T]J Am Heart Assoc.2016 Apr 19;5(4):e003350. doi: 10.1161/JAHA.116.003350. PMID: 27098970
7: Efficacy and Tolerability of Evolocumab vs Ezetimibe in Patients With Muscle-Related Statin Intolerance: The GAUSS-3 Randomized Clinical Trial.
[Nissen Steven E,Stroes Erik,Dent-Acosta Ricardo E,Rosenson Robert S,Lehman Sam J,Sattar Naveed,Preiss David,Bruckert Eric,Ceška Richard,Lepor Norman,Ballantyne Christie M,Gouni-Berthold Ioanna,Elliott Mary,Brennan Danielle M,Wasserman Scott M,Somaratne Ransi,Scott Rob,Stein Evan A,GAUSS-3 Investigators]JAMA.2016 Apr 19;315(15):1580-90. doi: 10.1001/jama.2016.3608. PMID: 27039291
8: Long-term effects following 4 years of randomized treatment with atorvastatin in patients with type 2 diabetes mellitus on hemodialysis.
[Krane Vera,Schmidt Kay-Renke,Gutjahr-Lengsfeld Lena J,Mann Johannes F E,März Winfried,Swoboda Florian,Wanner Christoph,4D Study Investigators (the German Diabetes and Dialysis Study Investigators)]Kidney Int.2016 Jun;89(6):1380-7. doi: 10.1016/j.kint.2015.12.033. Epub 2016 Feb 17. PMID: 26924051
9: Safety Profile of Atorvastatin 80 mg: A Meta-Analysis of 17 Randomized Controlled Trials in 21,910 Participants.
[Li Haixia,Wang Cailian,Zhang Shuo,Sun Sihao,Li Ruifei,Zou Meijuan,Cheng Gang]Drug Saf.2016 May;39(5):409-19. doi: 10.1007/s40264-016-0394-0. PMID: 26860922
10: Exposure to gemfibrozil and atorvastatin affects cholesterol metabolism and steroid production in zebrafish (Danio rerio).
[Al-Habsi Aziz A,Massarsky Andrey,Moon Thomas W]Comp Biochem Physiol B Biochem Mol Biol.2016 Sep;199:87-96. doi: 10.1016/j.cbpb.2015.11.009. Epub 2015 Nov 25. PMID: 26627126
11: Improvement in Renal Function and Reduction in Serum Uric Acid with Intensive Statin Therapy in Older Patients: A Post Hoc Analysis of the SAGE Trial.
[Deedwania Prakash C,Stone Peter H,Fayyad Rana S,Laskey Rachel E,Wilson Daniel J]Drugs Aging.2015 Dec;32(12):1055-65. doi: 10.1007/s40266-015-0328-z. PMID: 26625880
12: Multi-drug intoxication fatality involving atorvastatin: A case report.
[Cibickova Lubica,Caran Tomas,Dobias Martin,Ondra Peter,Vorisek Viktor,Cibicek Norbert]Forensic Sci Int.2015 Dec;257:e26-31. doi: 10.1016/j.forsciint.2015.09.020. Epub 2015 Oct 13. PMID: 26508377
13: Successful reintroduction of statin therapy after statin-associated rhabdomyolysis.
[Simons Janet E,Holbrook Anne M,Don-Wauchope Andrew C]J Clin Lipidol.2015 Jul-Aug;9(4):594-6. doi: 10.1016/j.jacl.2015.03.005. Epub 2015 Mar 28. PMID: 26228678
14: Ask the doctor. I am 61 and had been on atorvastatin for 10 years with no problems. Recently, I've had disabling muscle pain with both the generic atorvastatin and the brand-name version, Lipitor. My doctor says that I can no longer take statin drugs. Since strokes run in my family, I am concerned. Is there anything else I can do to decrease my risk of stroke?
[Fabiny Anne]Harv Womens Health Watch.2014 Nov;22(3):2. PMID: 26065097
15: Rhabdomyolysis precipitated by possible interaction of ticagrelor with high-dose atorvastatin.
[Kido Kazuhiko,Wheeler Mary B,Seratnahaei Arash,Bailey Alison,Bain Jonathan A]J Am Pharm Assoc (2003).2015 May-Jun;55(3):320-3. doi: 10.1331/JAPhA.2015.14151. PMID: 26003161
16: Protocols cure diseases, not patients: flaccid paresis in post-NSTEMI statin treatment.
[Rosada Javier,Rebelos Eleni,Petruccelli Stefania,Taddei Marco]BMJ Case Rep.2015 Apr 22;2015. pii: bcr2014208878. doi: 10.1136/bcr-2014-208878. PMID: 25903205
17: Statin adverse effects: sorting out the evidence.
[Mlodinow Steven G,Onysko Mary K,Vandiver Jeremy W,Hunter Melissa L,Mahvan Tracy D]J Fam Pract.2014 Sep;63(9):497-506. PMID: 25353029
18: Pemphigus erythematosus relapse associated with atorvastatin intake.
[Lo Schiavo Ada,Puca Rosa Valentina,Romano Francesca,Cozzi Roberto]Drug Des Devel Ther.2014 Sep 18;8:1463-5. doi: 10.2147/DDDT.S66666. eCollection 2014. PMID: 25258514
19: [Renoprotective efficacy of different doses of statins in experimental acute renal failure].
[Zeleniuk V H,Zamors'kyĭ I I,Horoshko O M]Fiziol Zh.2014;60(2):75-81. PMID: 25007525
20: [Rhabdomyolysis and severe hepatotoxicity due to a drug-drug interaction between ritonavir and simvastatin. Could we use the most cost-effective statin in all human immunodeficiency virus-infected patients?].
[Bastida Carla,Also Maria Antonia,Pericas Juan Manuel,Letang Emili,Tuset Montse,Miró Josep Maria]Enferm Infecc Microbiol Clin.2014 Nov;32(9):579-82. doi: 10.1016/j.eimc.2014.03.014. Epub 2014 Jun 7. PMID: 24913991
21: Statin-associated acute interstitial nephritis and rhabdomyolysis.
[Panchangam Vidyashankar]Saudi J Kidney Dis Transpl.2014 May;25(3):659-60. PMID: 24821172
22: Safety and tolerability of the use of atorvastatin 40 mg in common daily practice in short-term observation in 3,227 patients.
[Wiliński Jerzy,Dabrowski Mikołaj]Przegl Lek.2013;70(6):373-6. PMID: 24052972
23: Rhabdomyolysis developing secondary to atorvastatin therapy in a patient with liver cirrhosis.
[Eshraghian Ahad,Kamyab Amir A'lam]Intern Med.2013;52(7):823-5. Epub 2013 Apr 1. PMID: 23545684
24: Risk of hospitalized rhabdomyolysis associated with lipid-lowering drugs in a real-world clinical setting.
[Cziraky Mark J,Willey Vincent J,McKenney James M,Kamat Siddhesh A,Fisher Maxine D,Guyton John R,Jacobson Terry A,Davidson Michael H]J Clin Lipidol.2013 Mar-Apr;7(2):102-8. doi: 10.1016/j.jacl.2012.06.006. Epub 2012 Jul 3. PMID: 23415428
25: Severe colchicine intoxication in a renal transplant recipient on cyclosporine.
[Garrouste C,Philipponnet C,Kaysi S,Enache I,Tiple A,Heng A E]Transplant Proc.2012 Nov;44(9):2851-2. doi: 10.1016/j.transproceed.2012.09.028. PMID: 23146540
26: [Rhabdomyolysis following the coprescription of atorvastatin and fusidic acid].
[Gabignon C,Zeller V,Le Guyader N,Desplaces N,Lidove O,Ziza J-M]Rev Med Interne.2013 Jan;34(1):39-41. doi: 10.1016/j.revmed.2012.09.006. Epub 2012 Oct 24. PMID: 23102978
27: Atorvastatin-related rhabdomyolysis and acute renal failure in a genetically predisposed patient with potential drug-drug interaction.
[Marusic Srecko,Lisicic Ante,Horvatic Ivica,Bacic-Vrca Vesna,Bozina Nada]Int J Clin Pharm.2012 Dec;34(6):825-7. doi: 10.1007/s11096-012-9717-0. Epub 2012 Oct 18. PMID: 23076661
28: Statin-induced rhabdomyolysis and dermatomyositis: a rare combination.
[Cannon Cecilia S]JAAPA.2012 Aug;25(8):30, 32-3. PMID: 22928274
29: A fatal case of cutaneous adverse drug-induced toxic epidermal necrolysis associated with severe rhabdomyolysis.
[Noordally Sheik Oaleed,Sohawon Schoeb,Vanderhulst Julien,Duttmann Ruth,Corazza Francis,Devriendt Jacques]Ann Saudi Med.2012 May-Jun;32(3):309-11. doi: 10.5144/0256-4947.2012.309. PMID: 22588445
30: Genetic predisposition to atorvastatin-induced myopathy: a case report.
[Francesca Notarangelo M,Marziliano N,Antonietta Demola M,Pigazzani F,Guidorossi A,Angelica Merlini P,Ardissino D]J Clin Pharm Ther.2012 Oct;37(5):604-6. doi: 10.1111/j.1365-2710.2012.01337.x. Epub 2012 May 14. PMID: 22582980
31: Acute rhabdomyolysis caused by combination therapy with atorvastatin and warfarin.
[Mackay J W,Fenech M E,Myint K S]Br J Hosp Med (Lond).2012 Feb;73(2):106-7. PMID: 22504754
32: Rhabdomyolysis precipitated by a sitagliptin-atorvastatin drug interaction.
[Bhome R,Penn H]Diabet Med.2012 May;29(5):693-4. doi: 10.1111/j.1464-5491.2011.03502.x. PMID: 22023482
33: Hypernatremia due to rhabdomyolysis in a patient on statin.
[Victor Suma M,Gnanaraj Anand,Abraham Georgie,Sankardas Mullasari Ajit]Saudi J Kidney Dis Transpl.2011 Sep;22(5):1037-8. PMID: 21912045
34: Statins for acute ischemic stroke.
[Squizzato Alessandro,Romualdi Erica,Dentali Francesco,Ageno Walter]Cochrane Database Syst Rev.2011 Aug 10;(8):CD007551. doi: 10.1002/14651858.CD007551.pub2. PMID: 21833959
35: Atorvastatin metabolite measurements as a diagnostic tool for statin-induced myopathy.
[Skottheim Ine B,Bogsrud Martin P,Hermann Monica,Retterstøl Kjetil,Åsberg Anders]Mol Diagn Ther.2011 Aug 1;15(4):221-7. doi: 10.2165/11591830-000000000-00000. PMID: 21815705
36: Problems in dealing with very rare adverse effects of new anticancer drugs: the example of trabectedin.
[Grosso Federica,D'Incalci Maurizio]Tumori.2011 Mar-Apr;97(2):256. doi: 10.1700/667.7796. PMID: 21617728
37: Statin-associated rhabdomyolysis: is there a dose-response relationship?
[Holbrook Anne,Wright Mitchell,Sung Melani,Ribic Christine,Baker Steven]Can J Cardiol.2011 Mar-Apr;27(2):146-51. doi: 10.1016/j.cjca.2010.12.024. PMID: 21459261
38: [Rhabdomyolysis associated with atorvastatin combined with amiodarone and fluconazole].
[Franz C C,Bruggisser M,Krähenbühl S,Rätz Bravo A E]Praxis (Bern 1994).2011 Mar 2;100(5):273-84. doi: 10.1024/1661-8157/a000491. PMID: 21365557
39: Rhabdomyolysis following co-prescription of fusidic acid and atorvastatin.
[Teckchandani S,Robertson S,Almond A,Donaldson K,Isles C]J R Coll Physicians Edinb.2010 Mar;40(1):33-6. doi: 10.4997/JRCPE.2010.108. PMID: 21125037
40: Multiple organ failure after an overdose of less than 0.4 mg/kg of colchicine: role of coingestants and drugs during intensive care management.
[Montiel Virginie,Huberlant Vincent,Vincent Marie-Françoise,Bonbled Frédéric,Hantson Philippe]Clin Toxicol (Phila).2010 Oct;48(8):845-8. doi: 10.3109/15563650.2010.509101. Epub 2010 Aug 12. PMID: 20969505
41: Statins alter intracellular calcium homeostasis in malignant hyperthermia susceptible individuals.
[Metterlein T,Schuster F,Tadda L,Hager M,Roewer N,Anetseder M]Cardiovasc Ther.2010 Dec;28(6):356-60. doi: 10.1111/j.1755-5922.2010.00237.x. Epub 2010 Oct 19. PMID: 20955541
42: Severe rhabdomyolysis as a consequence of the interaction of fusidic acid and atorvastatin.
[Magee Ciara N,Medani Samar A,Leavey Sean F,Conlon Peter J,Clarkson Michael R]Am J Kidney Dis.2010 Nov;56(5):e11-5. doi: 10.1053/j.ajkd.2010.07.011. PMID: 20888103
43: Year two assessment of fenofibric acid and moderate-dose statin combination: a phase 3, open-label, extension study.
[Kipnes Mark S,Roth Eli M,Rhyne James M,Setze Carolyn M,Lele Aditya,Kelly Maureen T,Sleep Darryl J,Stolzenbach James C]Clin Drug Investig.2010;30(1):51-61. doi: 10.2165/11319800-000000000-00000. PMID: 19995098
44: Human skeletal muscle drug transporters determine local exposure and toxicity of statins.
[Knauer Michael J,Urquhart Bradley L,Meyer zu Schwabedissen Henriette E,Schwarz Ute I,Lemke Christopher J,Leake Brenda F,Kim Richard B,Tirona Rommel G]Circ Res.2010 Feb 5;106(2):297-306. doi: 10.1161/CIRCRESAHA.109.203596. Epub 2009 Nov 25. PMID: 19940267
45: Statins and fibrate target ClC-1 - from side effects to CLC pharmacology.
[Zdebik Anselm A]Br J Pharmacol.2009 Apr;156(8):1204-5. doi: 10.1111/j.1476-5381.2008.00083.x. PMID: 19751314
46: Rhabdomyolysis a result of azithromycin and statins: an unrecognized interaction.
[Strandell Johanna,Bate Andrew,Hägg Staffan,Edwards I Ralph]Br J Clin Pharmacol.2009 Sep;68(3):427-34. doi: 10.1111/j.1365-2125.2009.03473.x. PMID: 19740401
47: Synergistic action of statins and nitrogen-containing bisphosphonates in the development of rhabdomyolysis in L6 rat skeletal myoblasts.
[Nishiguchi Tomoko,Akiyoshi Takeshi,Anami Setsuko,Nakabayashi Toshikatsu,Matsuyama Kenji,Matzno Sumio]J Pharm Pharmacol.2009 Jun;61(6):781-8. doi: 10.1211/jpp.61.06.0011. PMID: 19505369
48: Impact of high-dose atorvastatin in coronary heart disease patients age 65 to 78 years.
[Koren Michael J,Feldman Theodore,Mendes Robert A]Clin Cardiol.2009 May;32(5):256-63. doi: 10.1002/clc.20448. PMID: 19452483
49: [Metabolic rhabdomyolysis during statin therapy].
[Vergely N,Leca V,Antoine J-C,Germain N,Khalfallah Y,Estour B]Rev Med Interne.2009 Aug;30(8):711-3. doi: 10.1016/j.revmed.2008.10.336. Epub 2009 May 5. PMID: 19419805
50: Efficacy and safety of ABT-335 (fenofibric acid) in combination with atorvastatin in patients with mixed dyslipidemia.
[Goldberg Anne C,Bays Harold E,Ballantyne Christie M,Kelly Maureen T,Buttler Susan M,Setze Carolyn M,Sleep Darryl J,Stolzenbach James C]Am J Cardiol.2009 Feb 15;103(4):515-22. doi: 10.1016/j.amjcard.2008.10.025. Epub 2008 Dec 26. PMID: 19195513
51: [Statin therapy and muscle disorders].
[Abel Tatjána,Fehér János]Orv Hetil.2009 Feb 8;150(6):261-3. doi: 10.1556/OH.2009.28520. PMID: 19179258
52: Should high creatine kinase discourage the initiation or continuance of statins for the treatment of hypercholesterolemia?
[Glueck Charles J,Rawal Bishal,Khan Naseer Ahmed,Yeramaneni Samrat,Goldenberg Naila,Wang Ping]Metabolism.2009 Feb;58(2):233-8. doi: 10.1016/j.metabol.2008.09.019. PMID: 19154957
53: Primary prevention of cardiovascular mortality and events with statin treatments: a network meta-analysis involving more than 65,000 patients.
[Mills Edward J,Rachlis Beth,Wu Ping,Devereaux Philip J,Arora Paul,Perri Dan]J Am Coll Cardiol.2008 Nov 25;52(22):1769-81. doi: 10.1016/j.jacc.2008.08.039. PMID: 19022156
54: The efficacy and safety of ezetimibe for treatment of dyslipidemia after heart transplantation.
[Crespo-Leiro M G,Paniagua M J,Marzoa R,Grille Z,Naya C,Flores X,Rodriguez J A,Mosquera V,Franco R,Castro-Beiras A]Transplant Proc.2008 Nov;40(9):3060-2. doi: 10.1016/j.transproceed.2008.09.007. PMID: 19010194
55: Relative safety profiles of high dose statin regimens.
[Escobar Carlos,Echarri Rocio,Barrios Vivencio]Vasc Health Risk Manag.2008;4(3):525-33. PMID: 18827903
56: Rhabdomyolysis with atorvastatin and fusidic acid.
[O'Mahony C,Campbell V L,Al-Khayatt M S,Brull D J]Postgrad Med J.2008 Jun;84(992):325-7. doi: 10.1136/pgmj.2007.064097. PMID: 18644925
57: Risk management of simvastatin or atorvastatin interactions with CYP3A4 inhibitors.
[Molden Espen,Skovlund Eva,Braathen Pia]Drug Saf.2008;31(7):587-96. PMID: 18558792
58: The creatine kinase response to eccentric exercise with atorvastatin 10 mg or 80 mg.
[Kearns Amy K,Bilbie Cherie L,Clarkson Priscilla M,White C Michael,Sewright Kim A,O'Fallon Kevin S,Gadarla Mamatha,Thompson Paul D]Atherosclerosis.2008 Sep;200(1):121-5. doi: 10.1016/j.atherosclerosis.2007.12.029. Epub 2008 Feb 7. PMID: 18261731
59: Oral nanoparticulate atorvastatin calcium is more efficient and safe in comparison to Lipicure in treating hyperlipidemia.
[Meena A K,Ratnam D Venkat,Chandraiah G,Ankola D D,Rao P Rama,Kumar M N V Ravi]Lipids.2008 Mar;43(3):231-41. doi: 10.1007/s11745-007-3142-5. Epub 2008 Jan 10. PMID: 18196308
60: Estimating the extent of reporting to FDA: a case study of statin-associated rhabdomyolysis.
[McAdams Mara,Staffa Judy,Dal Pan Gerald]Pharmacoepidemiol Drug Saf.2008 Mar;17(3):229-39. doi: 10.1002/pds.1535. PMID: 18175291
61: [Ishikawa diagram and barrier analysis implemented to a medication error that induces rhabdomyolysis].
[Hernández Martín J,Planells Herrero C,Font Noguera I,Poveda Andrés J L]Farm Hosp.2007 Sep-Oct;31(5):318-20. PMID: 18052637
62: CYP2D6*4 polymorphism is associated with statin-induced muscle effects.
[Frudakis Tony N,Thomas Matthew J,Ginjupalli Siva N,Handelin Barbara,Gabriel Richard,Gomez Hector J]Pharmacogenet Genomics.2007 Sep;17(9):695-707. PMID: 17700359
63: [Rhabdomyolysis associated to combined ezetimibe-statin treatment].
[Piedra León M,García Unzueta M T,Otero Martínez M,Amado Señaris J A]Rev Clin Esp.2007 Sep;207(8):425-6. PMID: 17688879
64: A multicentre, open study to assess the effect of individualizing starting doses of atorvastatin according to baseline LDL-C levels on achieving cholesterol targets: the Achieve Cholesterol Targets Fast with Atorvastatin Stratified Titration (ACTFAST-2) study.
[Farsang C,Athyros V,Gaw A,ACTFAST-2 investigators and Steering Committee members]Curr Med Res Opin.2007 Aug;23(8):1945-56. PMID: 17626713
65: Letter by Rosenberg and Uretsky regarding article, "Risks associated with statin therapy: a systematic overview of randomized clinical trials".
[Rosenberg Lauren,Uretsky Seth]Circulation.2007 Jul 3;116(1):e8; author reply e9. PMID: 17606852
66: Use of maximum-dose simvastatin or atorvastatin in an ethnically diverse population.
[Song Jessica C,Wang Clifford C,Huang Yi-Chao,Sanchez Jeannette,Nguyen Theresa,Khan Shamima,Kim Myo-Kyoung,Li Phoebe]Am J Health Syst Pharm.2007 Apr 1;64(7):767-72. PMID: 17384364
67: Atorvastatin-induced early-onset rhabdomyolysis in a patient with nephrotic syndrome.
[Jose Jimmy,Saravu Kavitha,Shastry Barkur Anathakrishna]Am J Health Syst Pharm.2007 Apr 1;64(7):726-9. PMID: 17384358
68: Safety of lovastatin/extended release niacin compared with lovastatin alone, atorvastatin alone, pravastatin alone, and simvastatin alone (from the United States Food and Drug Administration adverse event reporting system).
[Alsheikh-Ali Alawi A,Karas Richard H]Am J Cardiol.2007 Feb 1;99(3):379-81. Epub 2006 Dec 8. PMID: 17261402
69: [Rhabdomyolysis after treatment with atorvastatin detected by bone scintigraphy].
[Rubí S,Duch J,Ortín J,Setoain X,Pons F]Rev Esp Med Nucl.2006 Nov-Dec;25(6):393. PMID: 17173790
70: Risk factors for rhabdomyolysis with simvastatin and atorvastatin.
[Golomb Beatrice,Evans Marcella]Drug Saf.2006;29(12):1191; author reply 1191-2. PMID: 17147465
71: Risk factors for rhabdomyolysis with simvastatin and atorvastatin.
[Ronaldson Kathlyn J,O'Shea Justine M,Boyd Ian W]Drug Saf.2006;29(11):1061-7. PMID: 17061911
72: Effects of chronic treatment with statins and fenofibrate on rat skeletal muscle: a biochemical, histological and electrophysiological study.
[Pierno S,Didonna M P,Cippone V,De Luca A,Pisoni M,Frigeri A,Nicchia G P,Svelto M,Chiesa G,Sirtori C,Scanziani E,Rizzo C,De Vito D,Conte Camerino D]Br J Pharmacol.2006 Dec;149(7):909-19. Epub 2006 Oct 9. PMID: 17031388
73: Atorvastatin for stroke prevention.
Med Lett Drugs Ther.2006 Sep 11;48(1243):75-6. PMID: 16977287
74: Media report of rare rhabdomyolysis cases seems to have triggered reluctance among some New Zealanders to use statins.
[Gladding Patrick A,Kerr Andrew,Edwards Colin]N Z Med J.2006 Sep 8;119(1241):U2159. PMID: 16964305
75: Comparison of rosuvastatin versus atorvastatin in Hispanic-Americans with hypercholesterolemia (from the STARSHIP trial).
[Lloret Ramon,Ycas Joseph,Stein Michael,Haffner Steven,STARSHIP Study Group]Am J Cardiol.2006 Sep 15;98(6):768-73. Epub 2006 Jul 28. PMID: 16950182
76: Tolerability of atorvastatin in a population aged > or =65 years: a retrospective pooled analysis of results from fifty randomized clinical trials.
[Hey-Hadavi Judith H,Kuntze Erik,Luo Don,Silverman Paul,Pittman Donald,Lepetri Barbara]Am J Geriatr Pharmacother.2006 Jun;4(2):112-22. PMID: 16860258
77: [A case of interstitial lung disease with atorvastatin (Tahor) and a review of the literature about these effects observed under statins].
[Veyrac Gwenaëlle,Cellerin Laurent,Jolliet Pascale]Therapie.2006 Jan-Feb;61(1):57-67. PMID: 16792155
78: Quinine-induced renal failure as a result of rhabdomyolysis, haemolytic uraemic syndrome and disseminated intravascular coagulation.
[Lim A K H,Ho L,Levidiotis V]Intern Med J.2006 Jul;36(7):465-7. PMID: 16780456
79: Rhabdomyolysis in a patient treated with colchicine and atorvastatin.
[Tufan Abdurrahman,Dede Didem Sener,Cavus Safak,Altintas Neriman Defne,Iskit Alper Bektas,Topeli Arzu]Ann Pharmacother.2006 Jul-Aug;40(7-8):1466-9. Epub 2006 Jun 13. PMID: 16772404
80: Exposure of atorvastatin is unchanged but lactone and acid metabolites are increased several-fold in patients with atorvastatin-induced myopathy.
[Hermann Monica,Bogsrud Martin P,Molden Espen,Asberg Anders,Mohebi Beata U,Ose Leiv,Retterstøl Kjetil]Clin Pharmacol Ther.2006 Jun;79(6):532-9. PMID: 16765141
81: The statins: drug interactions of significance to the dental practitioner.
[Stevenson Helen,Longman Lesley P,Randall Christine,Field E Anne]Dent Update.2006 Jan-Feb;33(1):14-6, 18, 20. PMID: 16512094
82: Muscle symptoms associated with statins: a series of twenty patients.
[Soininen Kari,Niemi Mikko,Kilkki Esa,Strandberg Timo,Kivistö Kari T]Basic Clin Pharmacol Toxicol.2006 Jan;98(1):51-4. PMID: 16433891
83: Safety of statins when response is carefully monitored: a study of 336 heart recipients.
[Marzoa-Rivas R,Crespo-Leiro M G,Paniagua-Marin M J,Llinares-García D,Muñiz-Garcia J,Aldama-López G,Piñón-Esteban P,Campo-Pérez R,Castro-Beiras A]Transplant Proc.2005 Nov;37(9):4071-3. PMID: 16386629
84: Comparative safety of atorvastatin 80 mg versus 10 mg derived from analysis of 49 completed trials in 14,236 patients.
[Newman Connie,Tsai John,Szarek Michael,Luo Don,Gibson Eric]Am J Cardiol.2006 Jan 1;97(1):61-7. Epub 2005 Nov 15. PMID: 16377285
85: High-dose atorvastatin vs usual-dose simvastatin for secondary prevention after myocardial infarction: the IDEAL study: a randomized controlled trial.
[Pedersen Terje R,Faergeman Ole,Kastelein John J P,Olsson Anders G,Tikkanen Matti J,Holme Ingar,Larsen Mogens Lytken,Bendiksen Fredrik S,Lindahl Christina,Szarek Michael,Tsai John,Incremental Decrease in End Points Through Aggressive Lipid Lowering (IDEAL) Study Group]JAMA.2005 Nov 16;294(19):2437-45. PMID: 16287954
86: How common is rhabdomyolysis in patients receiving lipid-lowering therapy?
[Phillips Paul S]Nat Clin Pract Cardiovasc Med.2005 Mar;2(3):130-1. PMID: 16265453
87: Statin-induced apoptosis linked with membrane farnesylated Ras small G protein depletion, rather than geranylated Rho protein.
[Matzno Sumio,Yasuda Shinya,Juman Sachiko,Yamamoto Yukiko,Nagareya-Ishida Noriko,Tazuya-Murayama Keiko,Nakabayashi Toshikatsu,Matsuyama Kenji]J Pharm Pharmacol.2005 Nov;57(11):1475-84. PMID: 16259781
88: Changes in ubiquitin proteasome pathway gene expression in skeletal muscle with exercise and statins.
[Urso Maria L,Clarkson Priscilla M,Hittel Dustin,Hoffman Eric P,Thompson Paul D]Arterioscler Thromb Vasc Biol.2005 Dec;25(12):2560-6. Epub 2005 Oct 13. PMID: 16224050
89: Conversion to atorvastatin in patients intolerant or refractory to simvastatin therapy: the CAPISH study.
[Krasuski Richard A,Doeppenschmidt Dennis,Henry John S,Smith P Brad,Adinaro Joseph,Beck Rachel,Thompson Christopher M]Mayo Clin Proc.2005 Sep;80(9):1163-8. PMID: 16178496
90: Safety of high-dose atorvastatin therapy.
[Waters David D]Am J Cardiol.2005 Sep 5;96(5A):69F-75F. PMID: 16126026
91: Pharmacologic options for aggressive low-density lipoprotein cholesterol lowering: benefits versus risks.
[McKenney James M]Am J Cardiol.2005 Aug 22;96(4A):60E-66E. PMID: 16098846
92: Analysis of the global RNA expression profiles of skeletal muscle cells treated with statins.
[Morikawa Shigeru,Murakami Takeshi,Yamazaki Hiroyuki,Izumi Akashi,Saito Yasushi,Hamakubo Takao,Kodama Tatsuhiko]J Atheroscler Thromb.2005;12(3):121-31. PMID: 16020911
93: Intensive lipid lowering with atorvastatin in coronary disease.
[Ravnskov Uffe,Rosch Paul J,Sutter Morley C]N Engl J Med.2005 Jul 7;353(1):93-6; author reply 93-6. PMID: 16003834
94: Relative impact of CYP3A genotype and concomitant medication on the severity of atorvastatin-induced muscle damage.
[Wilke Russell A,Moore Jason H,Burmester James K]Pharmacogenet Genomics.2005 Jun;15(6):415-21. PMID: 15900215
95: Prevalence of potentially severe drug-drug interactions in ambulatory patients with dyslipidaemia receiving HMG-CoA reductase inhibitor therapy.
[Rätz Bravo Alexandra E,Tchambaz Lydia,Krähenbühl-Melcher Anita,Hess Lorenzo,Schlienger Raymond G,Krähenbühl Stephan]Drug Saf.2005;28(3):263-75. PMID: 15733030
96: A literature search on pharmacokinetic drug interactions of statins and analysis of how such interactions are reflected in package inserts in Japan.
[Saito M,Hirata-Koizumi M,Urano T,Miyake S,Hasegawa R]J Clin Pharm Ther.2005 Feb;30(1):21-37. PMID: 15659001
97: Exertion-induced rhabdomyolysis in a patient on statin therapy.
[Unnikrishnan Dilip,Satish B]Nephrol Dial Transplant.2005 Jan;20(1):244. PMID: 15632363
98: Rhabdomyolysis in a patient receiving atorvastatin and fluconazole.
[Kahri Juhani,Valkonen Miia,Bäcklund Tom,Vuoristo Matti,Kivistö Kari T]Eur J Clin Pharmacol.2005 Feb;60(12):905-7. Epub 2004 Dec 30. PMID: 15625612
99: The grapefruit challenge: the juice inhibits a crucial enzyme, with possibly fatal consequences.
[Karch Amy M]Am J Nurs.2004 Dec;104(12):33-5. PMID: 15621963
100: Potential for conflict of interest in the evaluation of suspected adverse drug reactions: use of cerivastatin and risk of rhabdomyolysis.
[Psaty Bruce M,Furberg Curt D,Ray Wayne A,Weiss Noel S]JAMA.2004 Dec 1;292(21):2622-31. Epub 2004 Nov 22. PMID: 15572720
101: Formulary conversion programs: the need for patient-specific risk assessment.
[Singh Sonal,Shrivastava Rakesh,Das Vijay]MedGenMed.2004 Sep 14;6(3):28. PMID: 15520652
102: Comparative pharmacokinetic interaction profiles of pravastatin, simvastatin, and atorvastatin when coadministered with cytochrome P450 inhibitors.
[Jacobson Terry A]Am J Cardiol.2004 Nov 1;94(9):1140-6. PMID: 15518608
103: Long-term safety and tolerability profile of ezetimibe and atorvastatin coadministration therapy in patients with primary hypercholesterolaemia.
[Ballantyne C M,Lipka L J,Sager P T,Strony J,Alizadeh J,Suresh R,Veltri E P]Int J Clin Pract.2004 Jul;58(7):653-8. PMID: 15311720
104: Rhabdomyolysis triggered by cytomegalovirus infection in a heart transplant patient on concomitant cyclosporine and atorvastatin therapy.
[Wong Wai-Man,Wai-Hung Shek Tony,Chan Koon-Ho,Chau Elaine,Lai Kam-Chuen]J Gastroenterol Hepatol.2004 Aug;19(8):952-3. PMID: 15242511
105: Atorvastatin decreases the coenzyme Q10 level in the blood of patients at risk for cardiovascular disease and stroke.
[Rundek Tatjana,Naini Ali,Sacco Ralph,Coates Kristen,DiMauro Salvatore]Arch Neurol.2004 Jun;61(6):889-92. PMID: 15210526
106: Clinical pharmacokinetics of atorvastatin.
[Lennernäs Hans]Clin Pharmacokinet.2003;42(13):1141-60. PMID: 14531725
107: Safety of atorvastatin derived from analysis of 44 completed trials in 9,416 patients.
[Newman Connie B,Palmer Gary,Silbershatz Halit,Szarek Michael]Am J Cardiol.2003 Sep 15;92(6):670-6. PMID: 12972104
108: Influence of 3-hydroxy-3-methylglutaryl-CoA (HMG-CoA) reductase inhibitors on endothelial nitric oxide synthase and the formation of oxidants in the vasculature.
[Parker Rex A,Huang Qi,Tesfamariam Belay]Atherosclerosis.2003 Jul;169(1):19-29. PMID: 12860247
109: Drug-induced rhabdomyolysis after concomitant use of clarithromycin, atorvastatin, and lopinavir/ritonavir in a patient with HIV.
[Mah Ming Jinell B,Gill M John]AIDS Patient Care STDS.2003 May;17(5):207-10. PMID: 12816614
110: Rhabdomyolysis causing AV blockade due to possible atorvastatin, esomeprazole, and clarithromycin interaction.
[Sipe Brooke E,Jones Ronald J,Bokhart Gordon H]Ann Pharmacother.2003 Jun;37(6):808-11. PMID: 12773066
111: Rhabdomyolysis and myalgia associated with anticholesterolemic treatment as potential signs of malignant hyperthermia susceptibility.
[Guis S,Bendahan D,Kozak-Ribbens G,Figarella-Branger D,Mattei J P,Pellissier J F,Treffouret S,Bernard V,Lando A,Cozzone P J]Arthritis Rheum.2003 Apr 15;49(2):237-8. PMID: 12687516
112: Treating dyslipidemia with statins: the risk-benefit profile.
[Clark Luther T]Am Heart J.2003 Mar;145(3):387-96. PMID: 12660659
113: Efficacy and safety of atorvastatin after pediatric heart transplantation.
[Chin Clifford,Gamberg Pat,Miller Joan,Luikart Helen,Bernstein Daniel]J Heart Lung Transplant.2002 Nov;21(11):1213-7. PMID: 12431495
114: The HMG Co-A reductase inhibitors ('statins') and myotoxic effects.
S Afr Med J.2002 Aug;92(8):596-7. PMID: 12244613
115: Rhabdomyolysis with concurrent atorvastatin and diltiazem.
[Lewin John J,Nappi Jean M,Taylor Marian H]Ann Pharmacother.2002 Oct;36(10):1546-9. PMID: 12243603
116: Pharmacological interactions of statins.
[Paoletti Rodolfo,Corsini Alberto,Bellosta Stefano]Atheroscler Suppl.2002 May;3(1):35-40. PMID: 12044584
117: Rhabdomyolysis with acute renal failure probably related to the interaction of atorvastatin and delavirdine.
[Castro Jose G,Gutierrez Luis]Am J Med.2002 Apr 15;112(6):505. PMID: 11959068
118: A study of the interaction potential of azithromycin and clarithromycin with atorvastatin in healthy volunteers.
[Amsden Guy W,Kuye Olatunde,Wei Greg C G]J Clin Pharmacol.2002 Apr;42(4):444-9. PMID: 11936570
119: Safety and efficacy of atorvastatin in heart transplant recipients.
[Patel Devang N,Pagani Francis D,Koelling Todd M,Dyke David B,Baliga Ragavendra R,Cody Robert J,Lake Kathleen D,Aaronson Keith D]J Heart Lung Transplant.2002 Feb;21(2):204-10. PMID: 11834348
120: Safety of HMG-CoA reductase inhibitors: focus on atorvastatin.
[Bernini F,Poli A,Paoletti R]Cardiovasc Drugs Ther.2001;15(3):211-8. PMID: 11713888
121: . . . from the 40th Interscience Conference on Antimicrobial Agents and Chemotherapy.
Res Initiat Treat Action.2000 Dec;6(4):28-31. PMID: 11708173
122: Atorvastatin: an updated review of its pharmacological properties and use in dyslipidaemia.
[Malhotra H S,Goa K L]Drugs.2001;61(12):1835-81. PMID: 11693468
123: Metabolism and drug interactions of 3-hydroxy-3-methylglutaryl coenzyme A-reductase inhibitors (statins).
[Igel M,Sudhop T,von Bergmann K]Eur J Clin Pharmacol.2001 Aug;57(5):357-64. PMID: 11599653
124: Acute rhabdomyolysis after atorvastatin and fusidic acid therapy.
[Wenisch C,Krause R,Fladerer P,El Menjawi I,Pohanka E]Am J Med.2000 Jul;109(1):78. PMID: 10991749
125: Short-term effect of atorvastatin in hypercholesterolaemic renal-transplant patients unresponsive to other statins.
[Romero R,Calviño J,Rodriguez J,Sánchez-Guisande D]Nephrol Dial Transplant.2000 Sep;15(9):1446-9. PMID: 10978406
126: New insights into the pharmacodynamic and pharmacokinetic properties of statins.
[Corsini A,Bellosta S,Baetta R,Fumagalli R,Paoletti R,Bernini F]Pharmacol Ther.1999 Dec;84(3):413-28. PMID: 10665838
127: Atorvastatin compared with simvastatin-based therapies in the management of severe familial hyperlipidaemias.
[Wierzbicki A S,Lumb P J,Semra Y,Chik G,Christ E R,Crook M A]QJM.1999 Jul;92(7):387-94. PMID: 10627888
128: Rhabdomyolysis associated with concomitant use of atorvastatin and cyclosporine.
[Maltz H C,Balog D L,Cheigh J S]Ann Pharmacother.1999 Nov;33(11):1176-9. PMID: 10573315
129: Safety of low-density lipoprotein cholestrol reduction with atorvastatin versus simvastatin in a coronary heart disease population (the TARGET TANGIBLE trial).
[März W,Wollschläger H,Klein G,Neiss A,Wehling M]Am J Cardiol.1999 Jul 1;84(1):7-13. PMID: 10404843
130: Judicious evaluation of adverse drug reactions: inaccurate assessment of 3-hydroxy-3-methylglutaryl coenzyme A reductase inhibitor-induced muscle injury.
[Dietz B L,Oberg K C]Pharmacotherapy.1999 Feb;19(2):232-5. PMID: 10030775
131: Atorvastatin in the treatment of primary hypercholesterolemia and mixed dyslipidemias.
[Yee H S,Fong N T]Ann Pharmacother.1998 Oct;32(10):1030-43. PMID: 9793596
132: Rhabdomyolysis after taking atorvastatin with gemfibrozil.
[Duell P B,Connor W E,Illingworth D R]Am J Cardiol.1998 Feb 1;81(3):368-9. PMID: 9468088
Disclaimer:
The content of this database of rhabdomyolysis is intended for educational and scientific research purposes only. It is not intended as a substitute for professional medical advice, diagnosis or treatment.
The views presented in this website do not necessarily reflect current or future opinion or policy of the US Food and Drug Administration. Any mention of commercial products is for clarification and not intended as endorsement.