Search for drugs:
Typing the drug name to query
ALISKIREN HEMIFUMARATE
DIR Classification
Classification:Less-DIR concern
Severity Score:1
Description in Drug Labeling: View Full Labeling: SPL in DailyMed | PDF
- ADVERSE REACTIONS
- Clinical Trials Experience
- Aliskiren's effect on ECG intervals was studied in a randomized, double-blind, placebo and active-controlled (moxifloxacin), 7-day repeat dosing study with Holter-monitoring and 12 lead ECGs throughout the interdosing interval. No effect of aliskiren on QT interval was seen.
Postmarketing Surveillance
Contingency Table:
Current Drug
Other Drugs
Rhabdomyolysis
18
42894
Other ADRs
8255
14109024
Odds Ratio = 0.718
Drug Property Information
ATC Code(s):
- C09DX02 - aliskiren hemifumarate
- C09DX -
- C09D - "ANGIOTENSIN II ANTAGONISTS, COMBINATIONS"
- C09 - AGENTS ACTING ON THE RENIN-ANGIOTENSIN SYSTEM
- C - CARDIOVASCULAR SYSTEM
- C09XA02 - aliskiren hemifumarate
- C09XA - Renin-inhibitors
- C09X - OTHER AGENTS ACTING ON THE RENIN-ANGIOTENSIN SYSTEM
- C09 - AGENTS ACTING ON THE RENIN-ANGIOTENSIN SYSTEM
- C - CARDIOVASCULAR SYSTEM
- C09XA52 - aliskiren hemifumarate
- C09XA - Renin-inhibitors
- C09X - OTHER AGENTS ACTING ON THE RENIN-ANGIOTENSIN SYSTEM
- C09 - AGENTS ACTING ON THE RENIN-ANGIOTENSIN SYSTEM
- C - CARDIOVASCULAR SYSTEM
- C09XA53 - aliskiren hemifumarate
- C09XA - Renin-inhibitors
- C09X - OTHER AGENTS ACTING ON THE RENIN-ANGIOTENSIN SYSTEM
- C09 - AGENTS ACTING ON THE RENIN-ANGIOTENSIN SYSTEM
- C - CARDIOVASCULAR SYSTEM
- C09XA54 - aliskiren hemifumarate
- C09XA - Renin-inhibitors
- C09X - OTHER AGENTS ACTING ON THE RENIN-ANGIOTENSIN SYSTEM
- C09 - AGENTS ACTING ON THE RENIN-ANGIOTENSIN SYSTEM
- C - CARDIOVASCULAR SYSTEM
Active Ingredient:aliskiren hemifumarate
Active Ingredient UNII:C8A0P8G029
Drugbank ID:DB09026
PubChem Compound:5493444
CAS Number:173334-57-1
Dosage Form(s):tablet, film coated
Route(s) Of Administrator:oral
Daily Dose:
- 150.0 mg/day C09XA02
Chemical Structure: 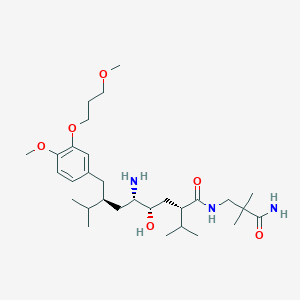

SMILE Code:
CC(C)[C@@H](CC1=CC(=C(C=C1)OC)OCCCOC)C[C@@H]([C@H](C[C@@H](C(C)C)C(=O)NCC(C)(C)C(=O)N)O)N
CC(C)[C@@H](CC1=CC(=C(C=C1)OC)OCCCOC)C[C@@H]([C@H](C[C@@H](C(C)C)C(=O)NCC(C)(C)C(=O)N)O)N
Reference
COHORT STUDY:
N/AOTHER REFERENCE(S):
N/ADisclaimer:
The content of this database of rhabdomyolysis is intended for educational and scientific research purposes only. It is not intended as a substitute for professional medical advice, diagnosis or treatment.
The views presented in this website do not necessarily reflect current or future opinion or policy of the US Food and Drug Administration. Any mention of commercial products is for clarification and not intended as endorsement.