Search for drugs:
Typing the drug name to query
DARUNAVIR
DIR Classification
Classification:Less-DIR concern
Severity Score:1
Description in Drug Labeling: View Full Labeling: SPL in DailyMed | PDF
- DRUG INTERACTIONS
- >>814301f9-c990-46a5-b481-2879a521a16f-1.jpeg
- >>814301f9-c990-46a5-b481-2879a521a16f-2.jpeg
- >>814301f9-c990-46a5-b481-2879a521a16f-3.jpeg
- >>814301f9-c990-46a5-b481-2879a521a16f-4.jpeg
- CLINICAL PHARMACOLOGY
- Pharmacodynamics
- Cardiac Electrophysiology
- In a thorough QT/QTc study in 40 healthy subjects, PREZISTA/ritonavir doses of 1.33 times the maximum recommended dose did not affect the QT/QTc interval.
Postmarketing Surveillance
Contingency Table:
Current Drug
Other Drugs
Rhabdomyolysis
31
42881
Other ADRs
3335
14113944
Odds Ratio = 3.06
Drug Property Information
ATC Code(s):
- J05AE10 - darunavir
- J05AE - Protease inhibitors
- J05A - DIRECT ACTING ANTIVIRALS
- J05 - ANTIVIRALS FOR SYSTEMIC USE
- J - ANTIINFECTIVES FOR SYSTEMIC USE
- J05AR14 - darunavir
- J05AR - "Antivirals for treatment of HIV infections, combinations"
- J05A - DIRECT ACTING ANTIVIRALS
- J05 - ANTIVIRALS FOR SYSTEMIC USE
- J - ANTIINFECTIVES FOR SYSTEMIC USE
Active Ingredient:darunavir ethanolate
Active Ingredient UNII:33O78XF0BW
Drugbank ID:DB01264
PubChem Compound:213039
CAS Number:206361-99-1
Dosage Form(s):suspension; tablet, film coated
Route(s) Of Administrator:oral
Daily Dose:
- 1200.0 mg/day J05AE10
Chemical Structure: 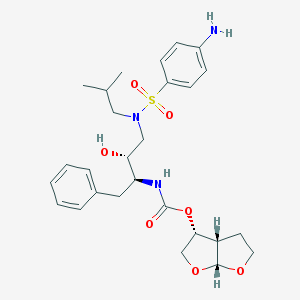

SMILE Code:
CC(C)CN(C[C@H]([C@H](CC1=CC=CC=C1)NC(=O)O[C@H]2CO[C@@H]3[C@H]2CCO3)O)S(=O)(=O)C4=CC=C(C=C4)N
CC(C)CN(C[C@H]([C@H](CC1=CC=CC=C1)NC(=O)O[C@H]2CO[C@@H]3[C@H]2CCO3)O)S(=O)(=O)C4=CC=C(C=C4)N
Reference
COHORT STUDY:
N/AOTHER REFERENCE(S):
1: No evident association between efavirenz use and suicidality was identified from a disproportionality analysis using the FAERS database.
[Napoli Andrew A,Wood Jennifer J,Coumbis John J,Soitkar Amit M,Seekins Daniel W,Tilson Hugh H]J Int AIDS Soc.2014 Sep 4;17:19214. doi: 10.7448/IAS.17.1.19214. eCollection 2014. PMID: 25192857
Disclaimer:
The content of this database of rhabdomyolysis is intended for educational and scientific research purposes only. It is not intended as a substitute for professional medical advice, diagnosis or treatment.
The views presented in this website do not necessarily reflect current or future opinion or policy of the US Food and Drug Administration. Any mention of commercial products is for clarification and not intended as endorsement.