Search for drugs:
Typing the drug name to query
DASATINIB
DIR Classification
Classification:Less-DIR concern
Severity Score:1
Description in Drug Labeling: View Full Labeling: SPL in DailyMed | PDF
- WARNINGS AND PRECAUTIONS
- QT Prolongation
- In vitro data suggest that dasatinib has the potential to prolong cardiac ventricular repolarization (QT interval). Of the 2440 patients with CML treated with SPRYCEL in clinical studies, 15 patients (<1%) had QTc prolongation reported as an adverse reaction. Twenty-two patients (1%) experienced a QTcF >500 ms. In 865 patients with leukemia treated with SPRYCEL in five Phase 2 single-arm studies, the maximum mean changes in QTcF (90% upper bound CI) from baseline ranged from 7.0 ms to 13.4 ms.
- Administer SPRYCEL with caution to patients who have or may develop prolongation of QTc. These include patients with hypokalemia or hypomagnesemia, patients with congenital long QT syndrome, patients taking anti-arrhythmic medicines or other medicinal products that lead to QT prolongation, and cumulative high-dose anthracycline therapy. Correct hypokalemia or hypomagnesemia prior to SPRYCEL administration.
- ADVERSE REACTIONS
- The following adverse reactions are discussed in greater detail in other sections of the labeling:
- Myelosuppression [see Dosage and Administration (2.3) and Warnings and Precautions (5.1)].
- Bleeding related events [see Warnings and Precautions (5.2)].
- Fluid retention [see Warnings and Precautions (5.3)].
- QT prolongation [see Warnings and Precautions (5.4)].
- Congestive heart failure, left ventricular dysfunction, and myocardial infarction [see Warnings and Precautions (5.5)].
- Pulmonary Arterial Hypertension [see Warnings and Precautions (5.6)].
- CLINICAL STUDIES
- Chronic Phase CML
- Dose-Optimization Study: A randomized, open-label study was conducted in patients with chronic phase CML to evaluate the efficacy and safety of SPRYCEL administered once daily compared with SPRYCEL administered twice daily. Patients with significant cardiac diseases, including myocardial infarction within 6 months, congestive heart failure within 3 months, significant arrhythmias, or QTc prolongation were excluded from the study. The primary efficacy endpoint was MCyR in patients with imatinib-resistant CML. A total of 670 patients, of whom 497 had imatinib-resistant disease, were randomized to the SPRYCEL 100 mg once daily, 140 mg once daily, 50 mg twice daily, or 70 mg twice daily group. Median duration of treatment was 22 months.
Postmarketing Surveillance
Contingency Table:
Current Drug
Other Drugs
Rhabdomyolysis
7
42905
Other ADRs
13600
14103679
Odds Ratio = 0.17
Drug Property Information
ATC Code(s):
- L01XE06 - dasatinib
- L01XE - Protein kinase inhibitors
- L01X - OTHER ANTINEOPLASTIC AGENTS
- L01 - ANTINEOPLASTIC AGENTS
- L - ANTINEOPLASTIC AND IMMUNOMODULATING AGENTS
Active Ingredient:dasatinib
Active Ingredient UNII:RBZ1571X5H
Drugbank ID:DB01254
PubChem Compound:3062316
CAS Number:302962-49-8
Dosage Form(s):tablet
Route(s) Of Administrator:oral
Daily Dose:
Chemical Structure: 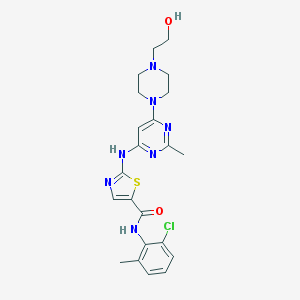

SMILE Code:
CC1=C(C(=CC=C1)Cl)NC(=O)C2=CN=C(S2)NC3=NC(=NC(=C3)N4CCN(CC4)CCO)C
CC1=C(C(=CC=C1)Cl)NC(=O)C2=CN=C(S2)NC3=NC(=NC(=C3)N4CCN(CC4)CCO)C
Reference
COHORT STUDY:
N/AOTHER REFERENCE(S):
1: An unexpected and devastating adverse event of dasatinib: Rhabdomyolysis.
[Uz Burak,Dolasik Ilhan]Leuk Res Rep.2015 Dec 3;5:1-2. doi: 10.1016/j.lrr.2015.11.001. eCollection 2016. PMID: 26870658
2: Reply: Management of De Novo CML and Imatinib-Induced Acute Rhabdomyolysis With the Second-Generation TKI, Dasatinib.
[Malkan Umit Y,Gunes Gursel,Etgul Sezgin,Aslan Tuncay,Aydin Seda,Haznedaroglu Ibrahim C]Ann Pharmacother.2016 Apr;50(4):336. doi: 10.1177/1060028016629981. Epub 2016 Feb 9. PMID: 26861992
3: Comment: Management of De Novo Chronic Myelogenous Leukemia and Imatinib-Induced Acute Rhabdomyolysis With the Second-Generation Tyrosine Kinase Inhibitor Dasatinib.
[Uz Burak,Dolasik Ilhan]Ann Pharmacother.2016 Apr;50(4):334-5. doi: 10.1177/1060028015627663. Epub 2016 Feb 2. PMID: 26839006
4: Management of De Novo CML and Imatinib-Induced Acute Rhabdomyolysis With the Second-Generation TKI, Dasatinib.
[Malkan Umit Yavuz,Gunes Gursel,Etgul Sezgin,Aslan Tuncay,Balaban Seda,Haznedaroglu Ibrahim C]Ann Pharmacother.2015 Jun;49(6):740-2. doi: 10.1177/1060028015579425. PMID: 25975995
Disclaimer:
The content of this database of rhabdomyolysis is intended for educational and scientific research purposes only. It is not intended as a substitute for professional medical advice, diagnosis or treatment.
The views presented in this website do not necessarily reflect current or future opinion or policy of the US Food and Drug Administration. Any mention of commercial products is for clarification and not intended as endorsement.