Search for drugs:
Typing the drug name to query
BREXPIPRAZOLE
DIR Classification
Classification:Most-DIR concern
Severity Score:4
Description in Drug Labeling: View Full Labeling: SPL in DailyMed | PDF
- CLINICAL PHARMACOLOGY
- Pharmacodynamics
- Cardiac Electrophysiology
- At a dose 3 times the MRHD for the treatment of schizophrenia and 4 times the MRHD for adjunctive therapy to antidepressants for the treatment of MDD, REXULTI does not prolong the QTc interval to any clinically relevant extent.
Postmarketing Surveillance
Contingency Table:
Current Drug
Other Drugs
Rhabdomyolysis
3
42909
Other ADRs
2615
14114664
Odds Ratio = 0.378
Drug Property Information
ATC Code(s):
- N05AX16 - brexpiprazole
- N05AX - Other antipsychotics
- N05A - ANTIPSYCHOTICS
- N05 - PSYCHOLEPTICS
- N - NERVOUS SYSTEM
Active Ingredient:brexpiprazole
Active Ingredient UNII:2J3YBM1K8C
Drugbank ID:DB09128
PubChem Compound:11978813
CAS Number:913611-97-9
Dosage Form(s):tablet
Route(s) Of Administrator:oral
Daily Dose:
- 3.0 mg/day N05AX16
Chemical Structure: 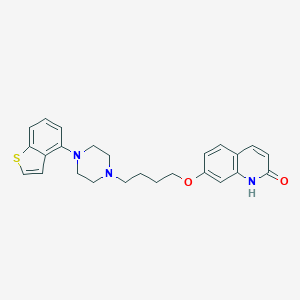

SMILE Code:
C1CN(CCN1CCCCOC2=CC3=C(C=C2)C=CC(=O)N3)C4=C5C=CSC5=CC=C4
C1CN(CCN1CCCCOC2=CC3=C(C=C2)C=CC(=O)N3)C4=C5C=CSC5=CC=C4
Reference
COHORT STUDY:
N/AOTHER REFERENCE(S):
N/ADisclaimer:
The content of this database of rhabdomyolysis is intended for educational and scientific research purposes only. It is not intended as a substitute for professional medical advice, diagnosis or treatment.
The views presented in this website do not necessarily reflect current or future opinion or policy of the US Food and Drug Administration. Any mention of commercial products is for clarification and not intended as endorsement.