Search for drugs:
Typing the drug name to query
ATAZANAVIR
DIR Classification
Classification:Moderate-DIR concern
Severity Score:3
Description in Drug Labeling: View Full Labeling: SPL in DailyMed | PDF
- WARNINGS AND PRECAUTIONS
- Cardiac Conduction Abnormalities
- Atazanavir has been shown to prolong the PR interval of the electrocardiogram in some patients. In healthy volunteers and in patients, abnormalities in atrioventricular (AV) conduction were asymptomatic and generally limited to first-degree AV block. There have been reports of second-degree AV block and other conduction abnormalities [see Adverse Reactions ( 6.2) and Overdosage ( 10) ]. In clinical trials that included electrocardiograms, asymptomatic first-degree AV block was observed in 5.9% of atazanavir-treated patients (n=920), 5.2% of lopinavir/ritonavir-treated patients (n=252), 10.4% of nelfinavir-treated patients (n=48), and 3.0% of efavirenz-treated patients (n=329). In Study AI424-045, asymptomatic first-degree AV block was observed in 5% (6/118) of atazanavir/ritonavir-treated patients and 5% (6/116) of lopinavir/ritonavir-treated patients who had on-study electrocardiogram measurements. Because of limited clinical experience in patients with preexisting conduction system disease (eg, marked first-degree AV block or second- or third-degree AV block). ECG monitoring should be considered in these patients. [See Clinical Pharmacology ( 12.2) .]
- DRUG INTERACTIONS
- >>bb717c4d-bd11-49af-8ec3-1efdce33a245-1.jpeg
- >>bb717c4d-bd11-49af-8ec3-1efdce33a245-2.jpeg
- >>bb717c4d-bd11-49af-8ec3-1efdce33a245-3.jpeg
- >>bb717c4d-bd11-49af-8ec3-1efdce33a245-4.jpeg
- ADVERSE REACTIONS
- Postmarketing Experience
- Cardiovascular System: second-degree AV block, third-degree AV block, left bundle branch block, QTc prolongation [see Warnings and Precautions ( 5.1) ]
- CLINICAL PHARMACOLOGY
- Pharmacodynamics
- Cardiac Electrophysiology
- Concentration- and dose-dependent prolongation of the PR interval in the electrocardiogram has been observed in healthy volunteers receiving atazanavir. In a placebo-controlled study (AI424-076), the mean (±SD) maximum change in PR interval from the predose value was 24 (±15) msec following oral dosing with 400 mg of atazanavir (n=65) compared to 13 (±11) msec following dosing with placebo (n=67). The PR interval prolongations in this study were asymptomatic. There is limited information on the potential for a pharmacodynamic interaction in humans between atazanavir and other drugs that prolong the PR interval of the electrocardiogram. [See Warnings and Precautions ( 5.1) .]
- Electrocardiographic effects of atazanavir were determined in a clinical pharmacology study of 72 healthy subjects. Oral doses of 400 mg (maximum recommended dosage) and 800 mg (twice the maximum recommended dosage) were compared with placebo; there was no concentration-dependent effect of atazanavir on the QTc interval (using Fridericia's correction). In 1793 HIV-infected patients receiving antiretroviral regimens, QTc prolongation was comparable in the atazanavir and comparator regimens. No atazanavir-treated healthy subject or HIV-infected patient in clinical trials had a QTc interval >500 msec. [See Warnings and Precautions ( 5.1) .]
Postmarketing Surveillance
Contingency Table:
Current Drug
Other Drugs
Rhabdomyolysis
44
42868
Other ADRs
5145
14112134
Odds Ratio = 2.816
Drug Property Information
ATC Code(s):
- J05AE08 - atazanavir
- J05AE - Protease inhibitors
- J05A - DIRECT ACTING ANTIVIRALS
- J05 - ANTIVIRALS FOR SYSTEMIC USE
- J - ANTIINFECTIVES FOR SYSTEMIC USE
- J05AR15 - atazanavir
- J05AR - "Antivirals for treatment of HIV infections, combinations"
- J05A - DIRECT ACTING ANTIVIRALS
- J05 - ANTIVIRALS FOR SYSTEMIC USE
- J - ANTIINFECTIVES FOR SYSTEMIC USE
Active Ingredient:atazanavir sulfate
Active Ingredient UNII:4MT4VIE29P
Drugbank ID:DB01072
PubChem Compound:148192
CAS Number:198904-31-3
Dosage Form(s):capsule, gelatin coated; powder
Route(s) Of Administrator:oral
Daily Dose:
- 300.0 mg/day J05AE08
Chemical Structure: 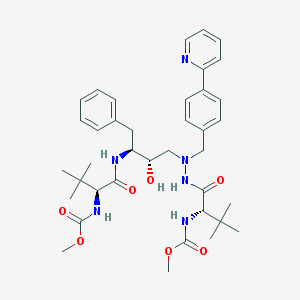

SMILE Code:
CC(C)(C)[C@@H](C(=O)N[C@@H](CC1=CC=CC=C1)[C@H](CN(CC2=CC=C(C=C2)C3=CC=CC=N3)NC(=O)[C@H](C(C)(C)C)NC(=O)OC)O)NC(=O)OC
CC(C)(C)[C@@H](C(=O)N[C@@H](CC1=CC=CC=C1)[C@H](CN(CC2=CC=C(C=C2)C3=CC=CC=N3)NC(=O)[C@H](C(C)(C)C)NC(=O)OC)O)NC(=O)OC
Reference
COHORT STUDY:
N/AOTHER REFERENCE(S):
1: Muscle symptoms and creatine phosphokinase elevations in patients receiving raltegravir in clinical practice: Results from the SCOLTA project long-term surveillance.
[Madeddu Giordano,De Socio Giuseppe V L,Ricci Elena,Quirino Tiziana,Orofino Giancarlo,Carenzi Laura,Franzetti Marco,Parruti Giustino,Martinelli Canio,Vichi Francesca,Penco Giovanni,Dentone Chiara,Celesia Benedetto Maurizio,Maggi Paolo,Libertone Raffaella,Bagella Paola,Di Biagio Antonio,Bonfanti Paolo,C.I.S.A.I. Group, Italy]Int J Antimicrob Agents.2015 Mar;45(3):289-94. doi: 10.1016/j.ijantimicag.2014.10.013. Epub 2014 Nov 13. PMID: 25476452
2: No evident association between efavirenz use and suicidality was identified from a disproportionality analysis using the FAERS database.
[Napoli Andrew A,Wood Jennifer J,Coumbis John J,Soitkar Amit M,Seekins Daniel W,Tilson Hugh H]J Int AIDS Soc.2014 Sep 4;17:19214. doi: 10.7448/IAS.17.1.19214. eCollection 2014. PMID: 25192857
3: Tolerability of HIV postexposure prophylaxis with tenofovir/emtricitabine and lopinavir/ritonavir tablet formulation.
[Tosini William,Muller Philippe,Prazuck Thierry,Benabdelmoumen Ghania,Peyrouse Eric,Christian Bernard,Quertainmont Yann,Bouvet Elisabeth,Rabaud Christian]AIDS.2010 Sep 24;24(15):2375-80. doi: 10.1097/QAD.0b013e32833dfad1. PMID: 20729709
4: Rhabdomyolysis in an HIV-infected patient on anti-retroviral therapy precipitated by high-dose pravastatin.
[Mikhail Nasser,Iskander Elizabeth,Cope Dennis]Curr Drug Saf.2009 May;4(2):121-2. PMID: 19442105
5: Severe rhabdomyolysis and acute renal failure secondary to concomitant use of simvastatin, amiodarone, and atazanavir.
[Schmidt Ginelle A,Hoehns James D,Purcell Jessica L,Friedman Robert L,Elhawi Yasir]J Am Board Fam Med.2007 Jul-Aug;20(4):411-6. PMID: 17615423
Disclaimer:
The content of this database of rhabdomyolysis is intended for educational and scientific research purposes only. It is not intended as a substitute for professional medical advice, diagnosis or treatment.
The views presented in this website do not necessarily reflect current or future opinion or policy of the US Food and Drug Administration. Any mention of commercial products is for clarification and not intended as endorsement.