Search for drugs:
Typing the drug name to query
ZILEUTON
DIR Classification
Classification:Moderate-DIQT concern
Severity Score:3.0
Description in Drug Labeling: View Full Labeling: SPL in DailyMed | PDF
- PRECAUTIONS
- Drug Interactions:
- In a drug interaction study in 16 healthy volunteers, co-administration of multiple doses of terfenadine (60 mg every 12 hours) and ZYFLO (600 mg every 6 hours) for 7 days resulted in a decrease in clearance of terfenadine by 22% leading to a statistically significant increase in mean AUC and C max of terfenadine of approximately 35%. This increase in terfenadine plasma concentration in the presence of ZYFLO was not associated with a significant prolongation of the QTc interval. Although there was no cardiac effect in this small number of healthy volunteers, given the high inter-individual pharmacokinetic variability of terfenadine, co-administration of ZYFLO and terfenadine is not recommended.
Postmarketing Surveillance
Contingency Table:
Current Drug
Other Drugs
QT Prolongation
0
24092
Other ADRs
0
38381587
Odds Ratio = N/A
Drug Property Information
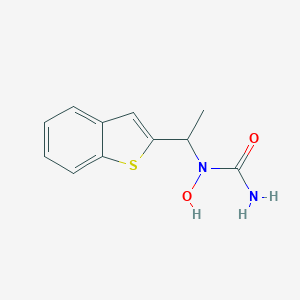
SMILE Code:
CC(N(O)C(N)=O)C1=CC2=CC=CC=C2S1
CC(N(O)C(N)=O)C1=CC2=CC=CC=C2S1
Reference
1: Drug interactions with cisapride: clinical implications.
[Michalets E L,Williams C R]Clin Pharmacokinet,2000 Jul;39(1):49-75. PMID: 10926350
Disclaimer:
The content of this database of QT prolongation is intended for educational and scientific research purposes only. It is not intended as a substitute for professional medical advice, diagnosis or treatment.
Any mention of commercial products is for clarification and not intended as endorsement.