Search for drugs:
Typing the drug name to query
DARIFENACIN
DIR Classification
Classification:Ambiguous
Severity Score:1.0
Description in Drug Labeling: View Full Labeling: SPL in DailyMed | PDF
- CLINICAL PHARMACOLOGY
- Electrophysiology
- The effect of six-day treatment of 15-mg and 75-mg ENABLEX on QT/QTc interval was evaluated in a multiple-dose, double-blind, randomized, placebo- and active-controlled (moxifloxacin 400 mg) parallel-arm design study in 179 healthy adults (44% male, 56% female) aged 18 to 65. Subjects included 18% PMs and 82% EMs. The QT interval was measured over a 24-hour period both predosing and at steady state. The 75-mg ENABLEX dose was chosen because this achieves exposure similar to that observed in CYP2D6 poor metabolizers administered the highest recommended dose (15 mg) of darifenacin in the presence of a potent CYP3A4 inhibitor. At the doses studied, ENABLEX did not result in QT/QTc interval prolongation at any time during the steady state, while moxifloxacin treatment resulted in a mean increase from baseline QTcF of about 7.0 msec when compared to placebo. In this study, darifenacin 15-mg and 75-mg doses demonstrated a mean heart rate change of 3.1 and 1.3 bpm, respectively, when compared to placebo. However, in the Phase II/III clinical studies, the change in median HR following treatment with ENABLEX was no different from placebo.
Postmarketing Surveillance
Contingency Table:
Current Drug
Other Drugs
QT Prolongation
2
24090
Other ADRs
4241
38377346
Odds Ratio = 0.752
Drug Property Information
ATC Code(s):
- G04BD10 - darifenacin
- G04BD - Urinary antispasmodics
- G04B - "OTHER UROLOGICALS, INCL. ANTISPASMODICS"
- G04 - UROLOGICALS
- G - GENITO URINARY SYSTEM AND SEX HORMONES
Active Ingredient:DARIFENACIN
Active Ingredient UNII:APG9819VLM
Drugbank ID:DB00496
PubChem Compound:444031
CTD ID:C101207
PharmGKB:PA164774901
CAS Number:133099-04-4
Dosage Form(s):tablet, extended release
Route(s) Of Administrator:oral
Daily Dose:
- 7.5 mg/day G04BD10
Chemical Structure: 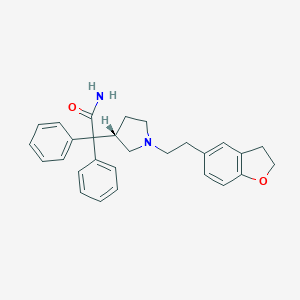

SMILE Code:
NC(=O)C([C@@H]1CCN(CCC2=CC3=C(OCC3)C=C2)C1)(C1=CC=CC=C1)C1=CC=CC=C1
NC(=O)C([C@@H]1CCN(CCC2=CC3=C(OCC3)C=C2)C1)(C1=CC=CC=C1)C1=CC=CC=C1
Reference
N/A
Disclaimer:
The content of this database of QT prolongation is intended for educational and scientific research purposes only. It is not intended as a substitute for professional medical advice, diagnosis or treatment.
Any mention of commercial products is for clarification and not intended as endorsement.